A b s t r a c t Kombucha is a fermented beverage made from brewed tea and sugar. The taste is slightly sweet and acidic and it may have residual carbon dioxide. Kombucha is consumed in many countries as a health beverage and it is gaining in popularity in the U.S. Consequently, many retailers and food service operators are seeking to brew this beverage on site. As a fermented beverage, kombucha would be categorized in the Food and Drug Administration model Food Code as a specialized process and would require a variance with submission of a food safety plan. This special report was created to assist both operators and regulators in preparing or reviewing a kombucha food safety plan.

Introduction
Kombucha is a fermented beverage made from brewed tea and sugar. The taste is slightly sweet and acidic and it may have residual carbon dioxide. Kombucha is consumed in many countries as a health beverage. It is believed to have prophylactic and therapeutic benefits toward a wide variety of ailments (Greenwalt, Steinkraus, & Ledford, 2000).
The kombucha process resembles a vinegar fermentation. Like vinegar, kombucha is a yeast fermentation of sugar to alcohol followed by a bacterial fermentation of alcohol to acetic acid. The symbiotic culture forms a pellicle or biofilm on the surface of the brew often called a mushroom or SCOBY (symbiotic culture of bacteria and yeast). The yeasts in the mixture metabolize sucrose into glucose and fructose, then into ethanol and carbon dioxide (Mayser, Fromme, Leitzmann, & Gründer, 1995). Ethanol is then oxidized by the bacteria (in the presence of air) to acetaldehyde, then to acetic acid (Mayser et al., 1995). Typically, the alcohol and acetic acid content of kombucha is less than 1%, respectively, but each can rise to 3% during a long ferment (~30 days; Mayser et al., 1995). The acetic acid bacteria also utilize glucose to produce gluconic acid to approximately 2%. Fructose is used to a lesser extent and some remains after the fermentation. Some glucose will remain unmetabolized, and together with the remaining fructose, provides sweetness.
As a fermented beverage, kombucha would be categorized in the Food and Drug Administration (FDA) model Food Code as a specialized process. A retail or food service operator would need to request a variance from their regulatory authority as defined in the Food Code sec- tion 3-502.11 (Food and Drug Administration [FDA], 2009). This section also requires that the operator submit a food safety plan to the regulatory authority for approval before commencing operations. Below is a hazard analysis and critical control points (HACCP)–based risk analysis of the process to help both operators and regulators maintain safe production of kombucha.
Process Flow
Naturally, kombucha recipes will vary. The general process has been described by Greenwalt and co-authors (2000) and includes infusing tea leaves (~4–5 g/L) into freshly boiled water. Sugar (sucrose) is added at 50–150 g/L (5% to 15%). The tea is allowed to brew for approximately 10 minutes and the tea leaves are removed. The tea is cooled to room temperature and approximately 100 ml/L (10%) of fresh-fermented kombucha containing the microbial mat from a previous batch is added to the sweetened tea. It is then covered with a clean porous cloth (e.g., cheese cloth) and incubated at room temperature for about 7–10 days. If the fermentation is allowed to continue beyond 10 days, acidity may rise to levels potentially harmful to consumers (equivalent to drinking undiluted vinegar).
Kombucha Hazards Analysis (Table 1)
Biological Hazards
Most boiling water with black or green tea infusions start at a pH of ≤5. Once fermentation starts that pH is reduced in approximately seven days to a nishing pH of ≤2.5. Fermentations such as wort to beer have a similar pH reduction during fermentation, although beer will be closer to pH 4. Since the initial infusion uses boiling water we consult Table A of section 1-201.10(B) (FDA, 2009) to determine if this is a potentially hazardous food (PHF). The tea infusion would not be a PHF if the pH were ≤4.6. Since the tea is heated but not packaged, however, it may be subject to contamination after cooling. Therefore, we must also consult Table B of section 1-201.10(B) (FDA, 2009) that requires a pH ≤4.2 to be a non- PHF. Since kombucha starts at a potentially hazardous pH (~5) and finnishes below 4.2 this process would require food safety monitoring to ensure safety. This is also confirmed in the Food Code under section 3-502.11 (FDA, 2009), where it requires a food safety plan for any process that uses acidi cation to make a PHF into a non-PHF.
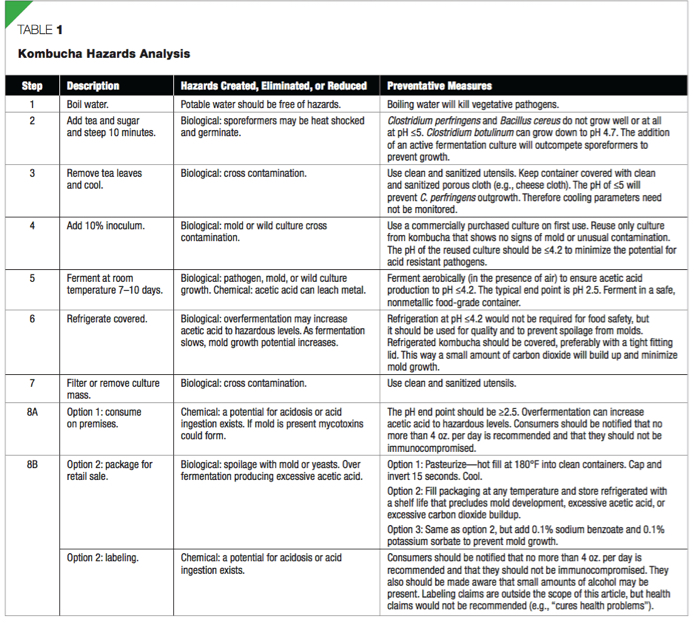
Chemical Hazards
FDA has evaluated the practices of several commercial producers of the kombucha and found no pathogenic organisms or other hygiene violations (Centers for Disease Control and Prevention [CDC], 1996). Kombucha consumption has proven to be harmful in only a few documented instances (Srinivasan, Smolinske, & Greenbaum, 1997). The possibility of toxic effects when kombucha is consumed in large quantities became a concern after two incidents in the U.S. in 1995. One individual died from perforations of the intestinal tract and severe acidosis. It was speculated that because she had recently increased her consumption threefold to 12 oz. that kombucha was the cause. The surviving victim mentioned that she increased the length of the fermentation time from 7 days to 14 days, and she could hardly manage swallowing the very acidic tea but did anyway. It was later determined that the individuals had severe preexisting conditions that made them susceptible to acidosis. These two cases of illness were investigated to determine if kombucha played a role in the development of metabolic acidosis or other toxic effects. It was concluded that kombucha is not harmful at about 4 oz. per day; however, potential risks are associated with excessive consumption or consumption by an individual with preexisting health problems (CDC, 1995).
Alcohol is certainly debatable as a hazard, but is not debatable as an impetus for taxes. Recently Severson (2010) reported some commercial producers of kombucha were forced to recall unpasteurized versions from grocery store shelves when the alcohol content exceeded 0.5%. Some brands continued to ferment and may produce up to 3% alcohol in the bottle. This happens because yeast continues to ferment sugars producing alcohol and carbon dioxide. In a closed container the buildup of carbon dioxide inhibits the conversion of alcohol to acetic acid.
Controlling Food Safety
Critical Control Points, Critical Limits, Monitoring, Corrective Actions, and Record Keeping
Of all of the steps in Table 1, only one is critical to prevent the potential for acid-resistant pathogens: step 5. In this step the fermentation proceeds from pH ~5 to ≤4.2. Therefore the critical limit is pH ≤4.2. The pH should be monitored using a calibrated digital pH meter for ease and accuracy (versus paper). The main corrective action if the pH > 4.3 would be to continue fermentation and remeasure. If the pH does not reach pH ≤4.2 in seven days the culture is most likely contaminated or the fermentation temperature is too cold. In this case discard is recommended. Start a new batch with a newly purchased commercial culture. A record of the pH of kombucha should be kept to verify that the safe pH level has been reached. An example would be to create a simple table (Table 2). For each batch, mark the start date (manufacture date) and starting pH, then mark each successive pH measurement until pH ≤4.2. You may optionally continue to measure pH since the operational target is 2.5. Operators will also need to keep a calibration log (Table 3). If the pH goes below 2.5 the operator can add fresh brewed tea to return it to pH ≥2.5. A pH measurement guide including calibration instructions can be found at
http://extension.usu.edu/files/publications/publication/FN_Food_Safety_2008-01.pdf

Good Manufacturing Practices
Controls and preventative measures that don’t meet the threshold of being critical, but nonetheless are needed to ensure safety, are contained in good manufacturing practices and standard operating procedures (SOPs). These controls are found in Table 1 under preventative measures. Using these preventa- tive measures is recommended
1. Use hot (≥165°F) water to steep tea (kills vegetative pathogens if present).
2. Use only clean and sanitary equipment and utensils.
3. Use a commercially purchased culture on first use. Reuse only culture from kombucha that shows no signs of mold or unusual contamination.
4.Kombucha with a pH of below 2.5 or that tastes especially acidic should not be offered to consumers. A corrective action would be to dilute the high acidity with fresh brewed tea until pH ≥ 2.5, but never higher than pH 4.2.
5. Discard all kombucha that is showing signs of mold contamination. Do not reuse for inoculum.
6. Consumer warnings: Consumers should be notified that no more than 4 oz. per day is recommended (see CDC references) and that they should not be immunocompromised. Furthermore, they should be made aware that minor amounts of alcohol may be present
7. Health claims: kombucha is suggested to offer health benefits. Operators are discouraged from marketing or labeling health claims such as drinking will “cure” some ailment.
SOPs
SOPs are written, step-by-step instructions to accomplish a food safety objective. The following are recommended.
1. A detailed pH measurement and calibration SOP.
2. A detailed process instruction sheet to tell employees how to make kombucha using the food safety measures outlined in this report. The SOP must describe how employees will measure and record on a pH log.
Retail Sale of Kombucha (Packaging)
Retail sale of kombucha is beyond the scope of this report. Described below, however, are some minimum concerns to this specialized process.
Many commercial processors bottle kombucha. The main food safety hazard is acid-resistant pathogens. Bottling kombucha at a pH ≤4.2 will ensure no pathogen growth. Another hazard is bottling an actively fermenting kombucha beverage. Carbon dioxide will build up inside the container causing pressure. As the pressure exceeds the ability of the container to hold it, leakage or breakage occurs. Bottles can explode, forming projectile hazards. The last concern is shelf life, where spoilage from mold can occur or alcohol can build up ≥0.5%. Typically, as a little carbon dioxide builds up, acetic acid production will cease. So an excess acid hazard is unusual.
Fermented beverages (foods) are exempt from acidied foods canning regulations. Therefore they do not need any filings or notications to FDA. Likewise, any refrigerated beverage (food) is also exempt from these same regulations.
Option 1: The best method is to pasteurize kombucha for bottling. Pasteurization will kill the culture preventing carbon dioxide or alcohol buildup in bottles. A simple recommendation is to heat kombucha to 180°F and bottle immediately. After 30 seconds invert bottle and hold for another 30 seconds. Allow bottles to cool. Pasteurized and bottled kombucha with a pH ≤ 4.2 is shelf stable (room temperature).
Option 2: This method relies on refrigeration and antifungal preservatives to minimize hazards and spoilage. Add 0.1% sodium benzoate and 0.1% potassium sorbate to kombucha with a pH ≤4.2. Bottle kombucha at any temperature. Keep refrigerated until use. Benzoate and sorbate will prevent mold growth and minimize yeast growth. Minimal to no growth of acetic acid bacteria will occur in bottles without significant oxygen. A refrigerated shelf life will need to be determined based on eventual yeast growth with carbon dioxide and alcohol production. If this proves difficult the operator may want to find a commercial kombucha culture with yeasts that do not grow well at refrigeration temperature.
Option 3: This method relies on refrigeration alone to minimize hazards and spoilage. Bottle kombucha at any temperature. Keep refrigerated until use. A refrigerated shelf life will need to be determined based on eventual yeast growth with carbon dioxide and alcohol production. If this proves difcult the operator may want to find a commercial kombucha culture with yeasts that do not grow well at refrigeration temperature.
Operators seeking to package kombucha for retail sale must also address labeling. Labeling issues are not covered here and operators are encouraged to inquire with their regulator or their state’s department of agriculture. Generally, regulators who over- see grocery (retail) have information on requirements for retail labels.
Questions and Answers
Can I make diet or low-sugar kombucha?
No. The sugar is required as part of the fermentation process. Without sugar no alcohol is produced and without alcohol no acetic acid can be produced.
comments (Ed) You can not Kombucha without sugar (great many sugars, fruits, juices, syrups, etc) but you can make kombucha that has a very low sugar content. As in any ferment the sugar undergoes transformation. How much sugar remains can be measured very simply by a Hydrometer or a Brix. These are routinely used by the industry such as the case of producing a Sweet Wine or a dry Wine. see our article on Bottling Kombucha.
I make kombucha using tea and other ingredients (e.g., fruit). Can I still use this guideline?
Yes, provided the added ingredient does not raise the starting pH over that of the original tea (~pH 5). It might still be safe to brew using higher starting pH levels, but the operator would need to consult a processing authority.
comments (Ed) I'm a little confused by the statement. .. "need to consult a processing authority". The known fact is that anything over a pH of 4.2 may be susceptible to mold forming. Especially if the pH remains high for several days. That does not mean that a ferment that starts high (pH > 5) can not produce a perfectly fine and safe brew (beer, wine, cider, kombucha, and pickles, kim-che, etc.). Additionally there are acids that one may add to the sugar and tea ("must" for beer & wine verbiage) that may be used to make the starting ferment lower. There is a real danger of bottling anything that has a pH >4.0 (according to State of Minnesota, Pickle Bill) as in the bottle even under refrigeration pathogens may develop.
Can I use this report as my HACCP Plan?
This report contains some of the information needed for an HACCP plan, just not in the typical table format. If the regulatory authority will accept it, it is suffcient. Copies of the recipe(s), a pH log, pH meter calibration log, and SOPs would need to be added to complete the food safety plan. If the kombucha is bottled, information on safe bottling and labels must also be included.
How strict is the recommendation from the CDC about limiting consumption of kombucha to 4 oz. per day?
The CDC recommendations are exactly that— recommendations. They are in response to two older and ill persons who became ill. One died. The analysis suggested they drank 12 oz. per day of very acidic kombucha. So, the recommendations are not to over ferment kombucha and limit servings.
I’ve read that the kombucha culture is sensitive to sanitizers. How can I sanitize utensils and wares so the culture is not harmed?
FDA model Food Code section 4-703.11 permits submerging previously cleaned wares and utensils in hot water (≥160°F) for ≥30 seconds. This will sanitize the wares and not leave any chemical residue.
comment (Ed) overall this is a much needed and respected outline for the kombucha enthusiast. Be they commercial, Farmers Market or Home Brewer. THank you Brian nummer for the outstanding work.
............................................................
HappyHerbalist.com online since 1997.
Kombucha Starter Cultures, Kombucha Prue Extract, Kombucha Mushroom Tea Concentrate, Homeopathic Preparation of Kombucha our Kombucha D1, pure Distilled Kombucha and Kombucha Herbal Extracts.
Ed Kasper was licensed by the State of California in the prescription of Traditional Chinese Medicine and developed Kombucha medicinal elixirs during his successful California Clinical Acupuncture & Herbal practice. We will be Happy to discuss your Kombucha concerns anytime. please email eddy@HappyHerbalist.com or text 919-518-3336 (EST)
References
Corresponding Author: Brian Nummer, Associate Professor, Nutrition, Dietetics and Food Sciences, Utah State University, 8700 Old Main Hill, Logan, UT 84322. E-mail: brian. nummer@usu.edu.
November 2013 • Journal of Environmental Health
Copyright 2013, National Environmental Health Association (www.neha.org).
Centers for Disease Control and Prevention. (1995). Unexplained severe illness possibly associated with consumption of kombu- cha tea—Iowa. Morbidity and Mortality Weekly Report, 44(48), 892–900.
Centers for Disease Control and Prevention. (1996). CDC editorial note. Journal of the American Medical Association, 275(2), 97–98.
Food and Drug Administration. (2009). Food code. Retrieved from http://www.fda.gov/Food/GuidanceRegulation/RetailFoodProtec- tion/FoodCode/UCM2019396.htm
Greenwalt, C.J., Steinkraus, K.H., & Ledford, R.A. (2000). Kombucha, the fermented tea: Microbiology, composition, and claimed health effects. Journal of Food Protection, 63(7), 976–981.
Mayser, P., Fromme, S., Leitzmann, C., & Gründer, K. (1995). The yeast spectrum of the “tea fungus kombucha.” Mycoses, 38(7–8), 289–295.
Severson, K. (2010). Kombucha may be treated like alcohol, government says. Retrieved from http://dinersjournal.blogs.nytimes.com/2010/ 06/25/kombucha-may-be-treated-like-alcohol-government-says/
Srinivasan, R., Smolinske S., & Greenbaum, D. (1997). Probable gastrointestinal toxicity of kombucha tea. Journal of General Inter- nal Medicine, 12(10), 643–644.

